“Equity” Stands A Chance In the International Health Regulations. Without Financing, Compliance At Stake [WG-IHR8]
Newsletter Edition #82 [Treaty Talks- IHR]
Hi,
When countries began sharing their visions to amend the International Health Regulations two years ago, there was strong resistance to broaden the scope of these technical regulations to fix what is missing - namely to reflect equity considerations, among others areas.
A persistent fight by developing countries to keep these issues on the table in these complex negotiations have paid off to a certain extent. This week countries are discussing proposals that could make this a reality. While we do not rule out realpolitik and tough bargaining to shape the final outcome, this is widely being seen as a significant development.
I am convinced individuals can make a difference even in the most polarized of times. The negotiations to amend the IHR illustrate what a clinical approach to conducting these discussions can accomplish while giving a nod to aspirations from countries. When the process is not jarring, discussions flow in a more predictable manner, albeit not fully insulated from the whirlwind of political considerations in rule-making.
Throwing journalistic staidness to the wind, I gushed to a senior official that the Bureau’s work explaining its rationale, read like music. (It is okay to do so once in a while!)
We hope you like this rather long edition. Thank you for reading.
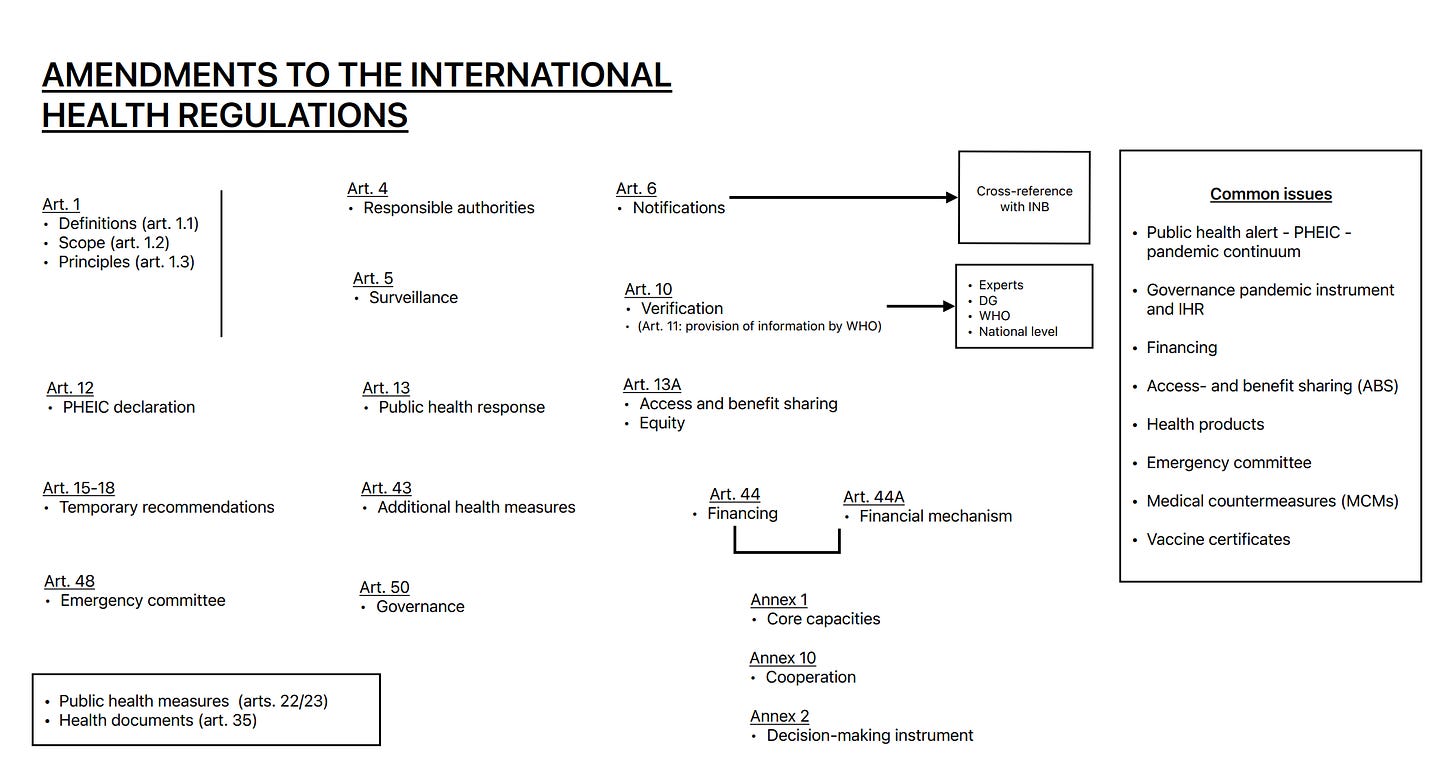
We also drew up a quick schematic - a snapshot of IHR provisions under consideration for amendments. You are welcome!
We are tracking this stuff for you in real-time. Help us meet reporting costs.
Support public interest global health journalism, become a paying subscriber. Tracking global health policy-making in Geneva is tough and expensive. Help us raise important questions, and in keeping an ear to the ground. Readers paying for our work makes this possible.
Our gratitude to our subscribers who help us stay in the game!
Until later.
Best,
Priti
Feel free to write to us: patnaik.reporting@gmail.com. Follow us on X: @filesgeneva
I. Analysis: WG-IHR8
“Equity” Stands A Chance In the International Health Regulations. Without Financing, Compliance At Stake
By Priti Patnaik & Tessa Jager
The Working Group set up to amend the International Health Regulations (WG-IHR) has tried to bell the cat. For the first time in more than two years of these negotiations, the Bureau of the WG-IHR has formulated proposals to reflect language that attempts to capture equity-related provisions suggested by developing countries in order to frame obligations on equitable access in the IHR. It however, shies away from pushing for a dedicated financing mechanism to ensure implementation of IHR obligations.
While this is a significant start, whether this beast can be tamed will be clear in the coming days. There is expected to be push back on some of these elements from developed countries and even some developing ones, who have long resisted the articulation of equity-related provisions in the IHR. Many believe that expanding the scope, is akin to violating the technical nature of these rules and succumbing to political considerations. But ultimately countries will decide what they want.
Reaching near-consensus this week will be key not only for the IHR, but also in setting the tone for the Pandemic Agreement negotiations which resume next week.
In a span of five days, countries are attempting to finalize the amendments to the International Health Regulations (2005). More than 300 proposals submitted by scores of countries have been distilled into targeted, and potentially far-reaching amendments, provided the negotiations this week proceed along the direction set out by the Bureau of the IHR. Last week, the WGIHR Bureau published the full text encompassing its proposals based on what countries have suggested. This is a marked shift towards transparency in a bid to fight forces of disinformation.
In a well-explained rationale, that has won broad support and appreciation, the Bureau provided context and reasoning, reflecting a careful balance of the technical and the political, in its proposed text to finalize the amendments. This rationale has not been posted publicly. Geneva Health Files has reviewed both the Bureau’s text and the accompanying rationale.
This story has two parts: we look at some key proposals from the Bureau’s text, followed by the rationale presented by the Bureau. We also review what’s on the table for the meeting underway this week. Finally, we present statements made non-state actors.
In his opening remarks at the final scheduled meeting of the WG-IHR, DG Tedros Adhanom Ghebreyesus urged countries to take more time if needed. He said:
“The text of the proposed amendments, for most of the articles and annexes, reflects the patience, flexibility and commitment you have towards reaching a consensus over the past year and a half of deliberations. In particular, I would like to commend your progress regarding the possible new designation of a pandemic emergency within the PHEIC process.
This week, I urge you to capitalize on this progress and to focus on the provisions related to equity and closing the gaps in the global health architecture that were exposed during the pandemic. I trust in the constructive spirit that has driven your negotiations. And I hope you approach this final stretch of your negotiations, keeping in mind the importance of maintaining the universality of the IHR.”
Co-Chairs Abdullah Asiri and Ashley Bloomfield kicked off the meeting.
Bloomfield said:
“Our objective is to finish the task of finalising a package of targeted amendments, while considering the need highlighted by the pandemic to take equity fully into account in the updated IHR. The Bureau is grateful for your support, constructive engagement, and flexibility to date, and our request is that we all work towards Friday this week, as a firm deadline.
We are all aware that the INB process remains live with another two weeks of intense negotiations scheduled following this meeting. We continue to work closely with the INB Co-Chairs and the Bureau to ensure our work is aligned. One reason it is important for us to complete our work this week, is so that there can be a full focus on the INB negotiations in the following two weeks, to maximize the chance of success in that crucial process. The ideal outcome, and I'm sure we are all striving for this, is for both processes to conclude successfully, to agreed texts, that are being adopted at the 77th World Health Assembly.
The Bureau is aware there may be a need for subsequent editorial tidy-up of the text that we will agree this week, and potentially to make small adjustments to reflect the outcome of the INB negotiation. We will seek your agreement before the end of the week to do this if necessary. And we would do so transparently and keep you fully informed. However, this should not preclude us getting agreement on the IHR amendments in this room, this week. And we have shaped our relevant proposals with a view to achieving this.
We should also recall that we are not rewriting the IHR to develop an IHR 2024. We are amending the IHR 2005, and they will continue to be reviewed and updated in the future, just as they have been previously amended in 2014 and 2022.
And finally, we must keep in the front of our minds this week, as the DG mentioned, just how important and urgent our work is. You will all be aware of the growing concern about the threat of H5H1 bird flu highlighted by the WHO just last week. We have the opportunity to ensure that the world is better prepared, both individual countries and collectively, to address that threat through strengthening core capacities in all States Parties. As Co-Chairs, we, and the Bureau, are here to help ensure that you can seize this opportunity.”

PART I
BUREAU’S PROPOSED TEXT TO AMEND THE IHR
Apart from significant provisions to reflect equity proposals from some developing countries, the Bureau has made suggestions on a vast area of technical matters including early action alerts; pandemic emergency; pathways and reasoning to declare the types of emergencies; use of personal data; status of health documents; responsibilities of the Director-General; and, interactions between State Parties and WHO in the context of an emergency. It also weighs on a “de-escalation” framework in the event of winding down of an emergency such as in the case of COVID-19.
(See Article 12 for example, on the determination of a public health emergency of international concern, including a pandemic emergency)
Note that these are just proposals pending negotiations, and endorsement from member states this week.
Below we review only few key features of the proposed text suggested by the Bureau (What we discuss below are excerpts from text and rationale. In bold, Bureau’s emphasis, not ours) .
Definitions
EARLY ACTION ALERT
Proposed text by Bureau:
“early action alert” means information and non-binding advice issued by the Director-General to States Parties on an event which, at the time of the consideration pursuant to paragraph 4 of Article 12, he or she has determined does not constitute a public health emergency of international concern.
Rationale:
“The Bureau considers that the global early warning and response functions, provided for by the current Regulations, should continue to revolve around the determination of PHEIC as their cornerstone, corresponding to the application of the proposed Bureau text for Article 12.
In particular, the Bureau notes that is important that an early action alert strengthens and improves the overall global early warning and response functions already provided for by the current Regulations.
· The Bureau considers that the introduction of an “early action alert”, –as per the proposed definition, i.e. a ‘by-default’ type of communication and advice to States Parties, issued by the Director-General when, pursuant to paragraph 4 of Article 12, an event is determined not to constitute a PHEIC, could provide a useful tool for the Director-General to: (i) raise awareness; and, (ii) provide technical advice to States Parties on preparedness and response actions vis-à-vis a given event even if does not constitute a PHEIC at that juncture. The issuance of an “early action alert”, inclusive of technical advice, could ultimately contribute to preventing the further evolution of an event into a PHEIC (or indeed pandemic emergency).
· Having considered the operational and procedural concerns expressed by some Member States with respect to the prospective introduction of an “early action alert”, for WHO to maintain the current level of operational and procedural agility, the Bureau considers that both the provision of specific advice by an IHR Emergency Committee about the issuance and content of an “early action alert”, as well as the issuance of related Temporary Recommendations, are not warranted. Thus, while the Director-General would be required to issue an “early action alert” if an event was assessed under Article 12(4) but determined not to constitute a PHEIC, the Director-General would determine advice contained in an “early action alert” that, similar to Temporary and Standing Recommendations, would be non-binding. The Bureau stresses that this would by no means prevent the Director-General from re-convening the Emergency Committee at a later stage if he or she believes the situation is warranting it.
……
- Thus, the inclusion of an “early action alert” does not impose a significant additional burden on the Director-General or the WHO Secretariat as, based on the implementation of the IHR to date, it is only likely to be issued every one to two years. (With respect to the operationalization of an “early action alert” and when such an alert would have been used (if it had existed) since the IHR (2005) came into effect the Bureau has reviewed the historic analysis provided by one Member States in writing. Expanding on it, since the IHR entered into effect in June 2007, to date (April 2024) “early action alerts” would have been issued on the following six occasions: MERS-CoV: 9 July 2013 (3 September 2015: last meeting of the IHR Emergency Committee; no PHEIC determined by the Director General); Yellow Fever, Angola and Democratic Republic of the Congo: 19 May 2016 onward (31 August 2016: second and last meeting of the IHR Emergency Committee; no PHEIC determined by the Director General); Ebola Virus Disease, Equateur Province, Democratic Republic of the Congo: from 18 May 2018 onward (18 May 2018: first and last meeting of the IHR Emergency Committee; no PHEIC determined by the Director General); Ebola Virus Disease, Kivu and Ituri Provinces, Democratic Republic of the Congo: from 17 October 2018 through 17 July 2019, when a PHEIC was determined; Mpox: 23 June through 23 July 2022, when a PHEIC was determined; COVID-19: from 23 through 30 January 2020, when a PHEIC was determined.
HEALTH PRODUCTS
Proposed text by Bureau:
“health products” means medicines; vaccines; medical devices including diagnostics; assistive products; vector control products, blood and other products of human origin. They are products used to prevent, protect, diagnose, treat diseases and health conditions.
Rationale:
“The Bureau was advised by the WHO Secretariat not to include a definition of “health technologies” in the IHR. The definition of “health technologies” contained in resolution WHA60.29 is regarded as too broad for the purpose of these Regulations and may cause unnecessary ambiguity.
Similarly, the Bureau was advised by the WHO Secretariat that the term “know-how” does not appear in any governing body resolutions, and that this term would create ambiguity if used as a technical term in the Regulations..
The Bureau notes that in the proposed Bureau text for the different Articles and Annes, the expression “health technologies” is not appearing and that reference to “technologies transfer” is made.”
PANDEMIC DEFINITION
Proposed text by Bureau:
“pandemic” means a public health emergency of international concern, that is infectious in nature and: (i) has spread and is spreading to and within multiple States Parties across WHO Regions; and (ii) is exceeding the capacity of health systems to respond in those States Parties; and (iii) is causing social and/or economic and/or political disruption in those States Parties; and (iv) requires rapid, equitable and enhanced coordinated international action, with whole[1]of-government and whole-of-society approaches.
PANDEMIC EMERGENCY DEFINITION
Proposed text by Bureau:
“pandemic emergency” means a public health emergency of international concern that is infectious in nature and: (i) is, or is likely to be, spreading to and within multiple States Parties across WHO Regions; and (ii) is exceeding, or is likely to exceed, the capacity of health systems to respond in those States Parties; and (iii) is causing, or is likely to cause, social and/or economic and/or political disruption in those States Parties; and (iv) requires rapid, equitable and enhanced coordinated international action, with whole[1]of-government and whole-of-society approaches.
Rationale:
“The proposed definition of “pandemic” is fully aligned with the definition of a “pandemic emergency” as noted in the rationale for “pandemic emergency”.
…The Bureau considers that the global early warning and response functions, provided for by the current Regulations, should continue to revolve around the determination of PHEIC as their cornerstone, corresponding to the application of the proposed Bureau text for Article 12.
· The Bureau considers that the introduction of a “pandemic emergency” event, as per the proposed definition, would represent a useful tool for the Director-General to characterize a type of PHEIC that has significant potential to become a pandemic.
· To that effect, the Bureau notes that when applying Article 12 and, by extension, Articles 48 and 49, the Director-General would also have to simultaneously and systematically consider whether a PHEIC also constitute a “pandemic emergency” and communicate to States Parties and to the public accordingly…..
· The definition of “pandemic emergency” proposed by the Bureau is based on suggestions received from Member States and makes limited use of qualifiers, as proposed by some Member States, to ensure clarity and minimise the risk of ambiguity. Additionally, since the determination of a “pandemic emergency” would demand the exercise of judgement by the Director-General, the limited use of qualifiers is also intended to limit any potential subsequent dispute surrounding the interpretation of such qualifiers.
· Considering the degree of judgement that the Director-General would have to apply when determining a “pandemic emergency”, the definition proposed by the Bureau does not deliberately quantify the four elements on which the proposed definition is built upon, e.g. specifying:
- the number of States Parties where the event has spread, or likely to be spreading; the number of WHO Regions where those States Parties are located; the number of States Parties, and levels (national vs proportion of sub-national administrative level/s) where the health system’s capacity to respond is exceeded or likely to be exceeded; the number of States Parties where the event is causing, or it is likely to cause, social/economic/political disruptions; degree of the social/economic/political disruptions caused by the event, or likely to be caused by it; what specific enhanced coordinated international action might be required, as this would vary depending on the circumstances.
…..The Bureau recognizes that, at the time when the Director-General makes the determination of “pandemic emergency”, all occurrences enumerated under numerals (i) through (iv) of the proposed definition of “pandemic emergency” may be current and actual. Under those circumstances, the event would, de facto, constitute a “pandemic”. While the Bureau regards the definition and determination of a “pandemic emergency” as adequate to shape and inform response actions under IHR provisions, as well as to trigger the application of any relevant provision of the prospective WHO Pandemic Agreement, the Bureau is also proposing to include, under Article 1, a definition of “pandemic”, with no related additional proposed amendments to the text of the IHR. This proposal by the Bureau responds to the request of the INB made at the joint WGIHR-INB meeting in July 2024 and is intended to also support coherence and alignment between the two instruments.
· Consistent with the suggestions received from Member States, the definition of “pandemic emergency” proposed by the Bureau does not include technical elements of the following nature: microbiological (e.g., novel or unknown pathogen; known pathogen having acquired novel characteristics); epidemiological (e.g., mode of transmission/novel mode of transmission); degree of susceptibility of the population worldwide; or clinical (e.g., morbidity and mortality). Nevertheless, the Bureau notes that such elements will be an integral part of a dynamic and reiterative event-specific risk assessment that the WHO Secretariat would be conducting, as well as the work of The Emergency Committee.
THE ACCESS TO HEALTH PRODUCTS
Proposed Text
Article 13 Public health response, including access to health products
1. Each State Party shall develop, strengthen and maintain, as soon as possible but no later than five years from the entry into force of these Regulations for that State Party, the core capacities to prepare for, and respond promptly and effectively to public health risks and public health emergencies of international concern, including a pandemic emergency, as set out in Annex 1. WHO shall publish, in consultation with Member States, guidelines to support States Parties in the development of public health response core capacities.
1.bis. Each State Party shall, within the means and resources at its disposal, provide sustainable domestic funding to build, strengthen and maintain the core capacities required under these Regulations.
2. Following the assessment referred to in paragraph 2, Part A of Annex 1, a State Party may report to WHO on the basis of a justified need and an implementation plan and, in so doing, obtain an extension of two years in which to fulfil the obligation in paragraph 1 of this Article. In exceptional circumstances and supported by a new implementation plan, the State Party may request a further extension not exceeding two years from the Director-General, who shall make the decision, taking into account the technical advice of the Review Committee. After the period mentioned in paragraph 1 of this Article, the State Party that has obtained an extension shall report annually to WHO on progress made towards the full implementation.
3. At the request of a State Party or following its acceptance of an offer by WHO, WHO shall collaborate in the response to public health risks and other events by providing technical guidance and assistance and by assessing the effectiveness of the control measures in place, including the mobilization of international teams of experts for on-site assistance, when necessary.
4. If WHO, in consultation with the States Parties concerned as provided in Article 12, determines that a public health emergency of international concern, including a pandemic emergency, is occurring, it may shall offer, in addition to the support indicated in paragraph 3 of this Article, further assistance to the State(s) Party(ies), to the extent possible, including an assessment of the severity of the international risk and the adequacy of control measures. Such collaboration may include the offer to mobilize international assistance in order to support the national authorities in conducting and coordinating on-site assessments. When requested by the State Party, WHO shall provide information supporting such an offer. The State Party shall make all efforts to reply to such an offer as soon as possible, including the rationale for the reply.
5. When requested by WHO, States Parties should shall provide, to the fullest extent possible within the means and resources at their disposal, support to WHO-coordinated response activities.
6. When requested, WHO shall provide appropriate guidance and assistance to other States Parties affected or threatened by the public health emergency of international concern, including a pandemic emergency.
7. WHO shall support States Parties and coordinate response activities during public health emergencies of international concern, including pandemic emergencies. To facilitate equitable access to health products, this support shall include, as necessary, coordinating with mechanisms and networks that facilitate equitable allocation and distribution of health products, including through technology transfer on mutually agreed terms. The aforesaid mechanisms and networks may include, but are not limited to, regional ones and those established under relevant international agreements.
8. After the determination of a public health emergency of international concern, pursuant to Article 12 of these Regulations, the Director-General shall:
(a) Conduct, and periodically review and update, an assessment of the availability and affordability of health products needed for the public health response; publish such assessment(s); and consider the assessment(s), when available, in connection to temporary recommendations pursuant to Articles 15, 17, 18, and 49 of these Regulations;
(b) make use of existing WHO-coordinated allocation mechanism(s) and networks, or facilitate their establishment as needed, to assist States Parties to access relevant health products based on public health risks and needs;
(c) support efforts by States Parties to scale-up and diversify, as appropriate, the production of relevant health products; (d) when requested by a State Party, share with the National Regulatory Authority of said State Party the dossier related to a specific health product, as provided to WHO by the manufacturer and where the manufacturer has consented, within 30 days from receiving such request for the purpose of facilitating regulatory authorization by the National Regulatory Authority;
(e) support States Parties, upon their request, to strengthen local production; achieve quality assurance through regulatory approval of locally manufactured products; and facilitate research and development and technology transfer on mutually agreed terms.
9. Pursuant to paragraph 5 of this Article, and paragraph 1 of Article 44 of these Regulations, States Parties shall, to the fullest extent possible, according to their national law and available resources, and upon request of other States Parties or WHO, undertake to collaborate with each other and to support WHO-coordinated responses activities, including through:
(a) supporting WHO in implementing actions outlined in paragraph 8 of this Article,
(b) engaging with, and encouraging, relevant non-State actors1 operating in their respective jurisdictions, to contribute towards equitable access to health products needed to respond to a public health emergency of international concern;
(c) publishing relevant terms of government-funded research agreements for health products needed to respond to a public health emergency of international concern, as well as information, where relevant, on pricing policies regarding these products and technologies, in order to support equitable access.
1 For the purpose of these Regulations, “non-State actors” shall be understood as defined in the WHO Framework of Engagement with non-State actors, adopted by the Sixty-ninth World Health Assembly through resolution WHA69.10 (2016), and any future amendment thereto.
Rationale
(New Paragraphs 7, 8 and 9):
“ The proposed new Paragraphs intend to operationalize the principles of equity and solidarity that the Bureau proposes to include in Article 2. They focus on the critical role of health products, and, where applicable, technology transfer, in response efforts.
· The Bureau considers that, for the overall economy of the text of the Regulations, aspects related to WHO-coordinated response activities, and to international response efforts in general, are more efficiently addressed under one single Article in a holistic manner. Therefore, the proposal is not to include a new article (Article 13A).
· As indicated in the Rationale related to the proposed definition “health products” under Article 1, the Bureau notes that “health products” are generally understood to constitute a sub-set of “health technologies” and that, given the extremely broad connotations of the expression “health technologies”, the inclusion of the definition of such expression for the purposes of these Regulations is not necessary.
FINANCING
Article 44 Collaboration and assistance, including financial assistance
Proposed Text:
1. States Parties shall undertake to collaborate with, and assist each other, to the fullest extent possible, in:
(a) the detection and assessment of, preparedness for, and response to, events as provided under these Regulations;
(b) the provision or facilitation of technical cooperation and logistical support, particularly in the development, strengthening and maintenance of the public health core capacities required under Annex 1 of these Regulations;
(c) the mobilization of financial resources to facilitate implementation of their obligations under these Regulations; and
(d) the formulation of proposed laws and other legal and administrative provisions for the implementation of these Regulations.; and
(e) the facilitation of access to health products, including through WHO-coordinated mechanisms.
2. WHO shall collaborate with, and assist, States Parties, upon request, to the extent possible, in:
(a) the evaluation and assessment of their public health core capacities in order to facilitate the effective implementation of these Regulations;
(b) the provision or facilitation of technical cooperation and logistical support to States Parties; and
(c) the mobilization of financial resources to support developing countries in building, strengthening and maintaining the capacities provided for in Annex 1.;
(d) strengthening domestic legislative and administrative arrangements for the implementation of these Regulations; and
(e) facilitating equitable access to health products through WHO-coordinated mechanisms.
2bis. Pursuant to Paragraph 1(c) of this Article, States Parties shall undertake to collaborate to:
(a) mobilize financial resources through all relevant sources, including existing and future bilateral, sub-regional, regional and multilateral funding mechanisms, to assist each other, in particular developing countries, as necessary and in complementarity to domestic funding as outlined in Paragraph 1bis of Article 13, in the implementation of these Regulations;
(b) encourage governance and operating models of existing financing entities and funding mechanisms to be responsive to the needs and national priorities, related to the these Regulations, of developing countries;
(c) secure the financial resources necessary to support the implementation of these Regulations through coordination and/or funding mechanisms that may be established in future International Agreement(s) related to pandemic prevention, preparedness and response;
(d) review the effectiveness of the provisions in this paragraph two years from their entry into force, and address identified gaps in financing IHR implementation that are not being met by current or future domestic funding, existing and new bilateral, sub-regional, regional and multilateral funding mechanisms, including, if necessary, through the establishment of a dedicated financing mechanism to provide targeted, supplementary financing, in particular to developing countries, to build, strengthen and maintain the capacities required under these Regulations.
2ter. The Director-General shall support the collaboration work in Paragraph 2bis above, as appropriate. The Parties and the Director-General shall report on its outcomes as part of the reporting to the Health Assembly.
3. Collaboration under this Article may be implemented through multiple channels, including bilaterally, through regional networks and the WHO regional offices, and through Intergovernmental organizations and international bodies.
Rationale:
“Paragraphs 1
· Chapeau: The Bureau considers that the addition of the term “fullest”, proposed by one Member States, further underscores the nature of the tangible commitment by States Parties to ensure that all States Parties - the “links” of the public health architecture - are equally strong.
….Letter (e): The Bureau considers that the proposed new Sub-paragraph - the content of which is elaborated upon in more detailed provisions proposed by the Bureau for Article 13 - is necessary to further emphasise the fact that health products constitute an area for which collaboration and assistance among States Parties are critical. The proposed new Sub-paragraphs ultimately intends to operationalize equity, calling on the solidarity among States Parties.
· Letter (e): The Bureau considers that the proposed new Sub-paragraph - the content of which is elaborated upon in more detailed provisions proposed by the Bureau for Article 13 - is necessary to further emphasize the fact that health products constitute an area for which collaboration and assistance are critical. The Bureau considers as pertinent the use of the adjective “equitable” in this proposed provision because WHO is ideally positioned to appraise and address its collaboration and assistance efforts to ensure the operationalization of equity. The proposed new Sub-paragraphs ultimately intends to operationalize equity, and implies that the principle of equity shall inspire and underpin any WHO initiative under this Regulations, including WHO-coordinated mechanisms).
Paragraph 2bis and 2ter
· The Bureau is proposing New Paragraphs 2bis and 2ter with the view to operationalize the principle of equity and solidarity.
· The Bureau developed this proposal taking into account the interactions among Member States in the context of the INB, as well as the presentations by the WHO Secretariat, during WGIHR meetings and intersessional briefings. In that light, the Bureau notes the significant number of existing financial mechanisms pertinent to the implementation of the Regulations. The Bureau notes the opportunity to make those mechanisms truly complementary and aligned so that they meet the IHR-related implantation needs of States Parties that require financial assistance The Bureau considers that this opportunity should be seized, with a view to a further review of the global funding and financing landscape vis-a-vis the status of domestic implementation of the Regulations in two years from the entry into force of the proposed amendment (Sub-paragraph 2bis.(d)). “
IMPLIMENTATION AND COMPLIANCE
Article 54bis: a “implementation and compliance committee”.
The committee is introduced under the “final arrangements” section of the IHR and therefore part of the governance provisions. It complements the Article 54 on reporting and review of the IHR, under which the following entities have the following obligations:
1) States Parties and the Director-General for reporting on the implementation of the IHR to the Health Assembly, without any timeframe imposed;
2) The Health Assembly for reviewing the implementation of the IHR (for the first time no later than 5 years after the adoption of the IHR), and
3) the WHO for reviewing and evaluating the functioning of Annex 2 at least once a year and submitting these reviews as appropriate to the Health Assembly for evaluation.
The proposed implementation and compliance committee would be a permanent committee that serves to assist States Parties to comply with their obligations under the IHR and strengthen core capacities. The text consolidated previous proposals for a new Article 53A, new Article 53 bis-quarter, Article 54 and new Article 54 bis, envisages the role of the Committee as both strengthening the steering role of States Parties in the implementation of the IHR and to provide a forum that explicitly addresses mutual accountability between States Parties and between States Parties and the WHO. The Committee would operationalize these tasks by firstly by “promoting and supporting learning, exchange of best practices, actions, and accountability among States Parties, at the national, regional and global levels’, and secondly through being “responsible for monitoring progress with IHR implementation, in order to support effective and equitable provision of technical assistance, logistical support and mobilization of financial resources…”.
The nature of the proposed Committee can be described as “facilitating”. It would function as a mechanism that helps to translate some of the objectives of the IHR, such as providing technical assistance and aiding collaboration between States Parties, into tangible action by reporting annually and making recommendations to the Health Assembly. In the current Bureau text, the Committee will consist of a to-be-decided amount of State Party Members from each WHO region that will serve for a to-be-decided amount of years. These State Parties will be represented by individuals with appropriate qualifications and experience. The text does not make clear what “appropriate” is and this leaves in the middle the extent to which the Committee members are assumed to be objective or subjective. However, as the text of the Article mentions that the Committee shall operate on the basis of consensus, and that States Parties that are not members of the IHR Implementation and Compliance Committee have a right to speak in Committee meetings, the text seems to assume a political background of Committee Members.
The current text also does not explicate a method of work for the Committee. While it is difficult to imagine the Committee can make recommendations to improve the implementation of the IHR by a State Party without assessing the status of the implementation, the current text does not mention that the Committee has a sort of “checking” or “overseeing” function. However, it does set out the principles in according to which it should work, are that it shall function in a manner that is “transparent, non-adversarial and non-punitive.”
FOOTNOTE ON PREQUALIFICATION
In an analysis, Third World Network drew attention to the proposed Bureau Text on a footnote to Annex 6 (on vaccination, prophylaxis and related certificates), that experts say “effectively neutralises equitable access especially through local production.”
Paragraph 3 of Annex 6 states “Certificates under this Annex are valid only if the vaccine or prophylaxis used has been approved by WHO”. The proposed footnote to this Para now states:
“Approved by WHO refers to health products evaluated and listed by WHO under the WHO Prequalification (PQ) procedures, applicable to medicines, vaccines, in-vitro diagnostics, vector control products, immunisation devices; and inspection services; and Emergence Use Listing (EUL) procedures, applicable to medicines, vaccines, in-vitro diagnostics”.
According to TWN:
“…A similar footnote is also proposed to the model international vaccination and prophylaxis certificate, which is part of Annex 6. This means that vaccination, prophylaxis or related certificate under the IHR will be recognised for international travel and related purpose only when the product used is prequalified by WHO.
This restricts the number of products which can be used for diagnosis, vaccination, and treatment. The insistence of WHO pre-qualified products would result in concentration and monopoly. Further, the compliance cost of WHO pre-qualification is high and only a few manufacturers from developing countries can meet the WHO pre-qualification standards. The WHO pre-qualification process also systematically discriminates against the regulatory approval by developing country manufacturers. As per the current practice, it is almost an automatic approval for products approved by WHO Listed authorities formerly known as stringent regulatory authorities. If an independent WHO evaluation is required for products approved by regulatory authorities, this would require time and resources.”
NOTABLE OMISSIONS
A. The Bureau’s text is not in favor of reflecting Common but Differentiated Responsibilities.
See rationale:
“…With respect to the proposed addition of the principle of “common but differentiated responsibility”, the Bureau recognizes that there are differing views on the inclusion of this terminology in the IHR. On balance, the Bureau considers it is critical to preserve the spirit and principle of universality underpinning the Regulations and, by extension, the right to health of all persons. Through the introductions of deadlines for the establishment of core capacities, pursuant to Articles 5, 13, and 19, the Regulations directly recognize the different epidemiological and socio-economic circumstances experienced by different States Parties. A similar recognition is also explicit in Articles 13 and 44, directly or indirectly related to the provision of assistance, and, most importantly in paragraph 3 of Article 59, regarding the adjustments of domestic legislative and administrative arrangements. Finally, a number of Bureau proposals in other Articles recognize and seek to address the different capacities and resources available to States Parties in implementing these Regulations, including by strengthening the obligations on developed States Parties and WHO to better support developing States Parties”
B. ACCESS AND BENEFITS SHARING
TWN points out that the Bureau’s text fully ignores the sharing proposal made by the Africa Group.
“The Africa Group had proposed the insertion of the following paragraph to Article 6 of IHR 2005:
“No sharing of genetic sequence data or information shall be required under these Regulations. The sharing of genetic sequence data or information shall only be considered after an effective and transparent access and benefit sharing mechanism with standard material transfer agreements governing access to and use of biological material including genetic sequence data or information relating to such materials as well as fair and equitable sharing of benefits arising from their utilization is agreed to by WHO Member States, is operational and effective in delivering fair and equitable benefit sharing”.
The absence of a fair and equitable benefit-sharing mechanism under IHR 2005 undermines equity by giving a free-riding possibility to pharmaceutical companies especially those based in developed countries to make use of the pathogen samples or their sequence information without any corresponding obligation to share the benefits emanating from their research and development. Though there was discussion to include the Pandemic Access and Benefit Sharing (PABS) system to include benefit sharing during PHEIC the latest INB Bureau’s text limits the benefit sharing only to the duration of the outbreak of a pandemic. This means there would not be any obligation to share benefits by contributing health products during a PHEIC. This goes against the mandate of addressing equity under decision EB 150.3.”
In the current H5N1 outbreak, the U.S. is facing criticism for not sharing enough information. (See Stat News: USDA faulted for disclosing scant information about outbreaks of H5N1 avian flu in cattle)
PART II
WGIHR8 – APRIL 22-26
In an agenda published ahead of the meeting, the Bureau detailed the issues that are being considered this week. Apart from discussing specific provisions, building on the work of previous meetings, a draft resolution text which will be considered at the World Health Assembly will also be discussed this week.
Some experts are of the view that both the INB and IHR processes should have different resolutions, to be considered by the Assembly given the lack of sufficient and uncertain progress in the INB track.
While some developing countries, including those from the Africa Group have maintained that both processes are tied, at this stage it appears more countries are keen to wrap up on the IHR negotiations in order to build momentum in these reforms discussions. Some diplomats also point to the immediacy and binding nature of the IHR, given its opt-out status, meaning that amendments will come into force unless countries choose not to do so within a specific time period. (The IHR have been adopted under Article 21 of the WHO Constitution.)
“The IHR are existing rules. We should wrap up these negotiations, also in the context of on-going emergencies”, a developing country diplomat told us this week.
Not all agree. Some believe that this process can be used as a leverage for making progress in the INB.
“Financing is a key issue for both processes. Without clarity on financing, we cannot accept greater implementation and compliance, even within IHR,” a small developing country negotiator said.
Some believe that if countries get to consensus broadly on the Bureau’s suggested text on the amendments to the IHR – there might even be lesser need for a Pandemic Agreement.
As for a new mechanism on Pathogen Access and Benefits Sharing, such rules can be brought in independently.
STATEMENTS BY NON-STATE ACTORS (Excerpts)
TWN:
“First, while we welcome the proposed language on WHO’s role in equitable access to health products, specific methods for achieving this remain absent, particularly in Para 7 of Article 13. Para 8, though detailing DG actions, lacks clarity on whether these actions are continuous or only post-PHEIC declaration. There are existing programmes which serves equity which the WHO Secretariat runs without a specific focus on emergencies and without legal certainty of delivery, for example Pharmacopeia. Therefore, we request to adopt certain language with Article 13, which makes these programmes accountable to States Parties.
Second, the inclusion of a footnote in Annex 6, limiting scope to WHO-prequalified products, could severely restrict the diversity of health product production, as highlighted by adverse consequences of PreQ noted in US ITC and ACT-A evaluations.
Third, Article 44 lacks provisions for an accountable financial mechanism. Feedback from external funding agencies suggests a reluctance to adhere to directives from WHO or IHR States Parties, emphasizing the need for a comprehensive, non-donor-driven funding mechanism.
Fourth, amendments proposed in Article 4 para 2 bis and Article 13 Para 1 bis shift the implementation burden onto State parties, contradicting the Common but Differentiated Responsibilities (CBDR) principle and abandoning support for developing countries. We also request expanding communication channels maintained in Article 4 to include communications under Article 13. Currently it is limited to communications upto Article 12.
Fifth, we caution against proposals for an early action alert system that shares information without adequate risk assessment, potentially causing panic and discriminatory responses as seen during the Omicron outbreak.”
IFPMA
“…we have three key concerns that could lead to uncertainty and would benefit from clear science- and evidence-based criteria to avoid politicization:
The proposed definitions of “Early Action Alerts,” “Public Health Emergency of International Concern (PHEIC), including a Pandemic Emergency,” and “Pandemics” as well as references to “health products” (including “relevant health products”) are excessively vague. This makes it very difficult for industry to assess the overall instrument.
The decision-making process to determine and make recommendations on “Early Action Alerts” and “PHEIC, including a Pandemic Emergency” leaves room for discretion and interpretation when it should be based on science- and evidence-based criteria.
There are recommendations in Article 13 that pre-empt the outcomes of the INB to improve equitable access, in particular the WHO-coordinated mechanisms and networks, and do not strike a balanced approach. It would be premature and potentially counterproductive to include it in IHR.”
Knowledge Ecology International
“In relation to the proposed Bureau’s text, published on April 17th, we welcome the transparency mandate contained in Article 13.9(c) which calls upon state parties to publish “relevant terms of government-funded research agreements for health products needed to respond to a public health emergency of international concern, as well as information, where relevant, on pricing policies regarding these products and technologies, in order to support equitable access.
Article 13.7 envisions that WHO plays a coordinating role among State Parties during public health emergencies of international concern. This coordinating role involves the facilitation of equitable access to health products, “including through technology transfer on mutually agreed terms.”
An important challenge in creating an enabling environment to facilitate technology transfer and know-how is to find ways to share access to the inventions, know-how and other inputs to drugs, vaccines and other countermeasures.
One approach is to create incentives for parties to share some rights acquired from publicly funded R&D or procurements, in a reciprocal manner, with parties that also share. Another approach would be to provide money or other incentives to acquire rights to patented inventions, know-how and other inputs, from private rights holders.
Did a colleague forward this edition to you? Sign up to receive our newsletters & support Geneva Health Files!
Global health is everybody’s business. Help us probe the dynamics where science and politics interface with interests. Support investigative global health journalism.